All AbMole products are for research use only, cannot be used for human consumption.
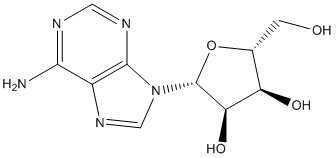
Adenosine is a nucleoside composed of a molecule of adenine attached to a ribose sugar molecule (ribofuranose) moiety via a β-N9-glycosidic bond. Extracellular adenosine concentrations from normal cells are approximately 300 nM; however, in response to cellular damage (e.g. in inflammatory or ischemic tissue), these concentrations are quickly elevated (600–1,200 nM). Thus, in regard to stress or injury, the function of adenosine is primarily that of cytoprotection preventing tissue damage during instances of hypoxia, ischemia, and seizure activity. Activation of A2A receptors produces a constellation of responses that in general can be classified as anti-inflammatory.

Neuropharmacology. 2025 Oct 22; .
Extracellular adenosine in acute stress-induced depressive-like behavior: Where does it come from?
Adenosine purchased from AbMole
| Molecular Weight | 267.24 |
| Formula | C10H13N5O4 |
| CAS Number | 58-61-7 |
| Solubility (25°C) | DMSO 12 mg/mL |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
[1] Shuli Man, et al. Potential and promising anticancer drugs from adenosine and its analogs
[2] Robert W Greene, et al. The adenosine-mediated, neuronal-glial, homeostatic sleep response
[3] J Novotn. Adenosine and its role in physiology
[4] Mehdi Kazemzadeh-Narbat, et al. Adenosine-associated delivery systems
| Related Metabolite/Endogenous Metabolite Products |
|---|
| 2'-Deoxyinosine
2′-Deoxyinosine is a nucleoside composed of hypoxanthine attached to 2′-deoxyribose via a β-N9-glycosidic bond. It is a DNA damage product resulting from the impairment of DNA by reactive nitrogen species. 2′-deoxyinsine can be used as a model compound to study the chemistry of adduct formation and radical chemistry that may affect DNA structures. 2′-Deoxyinosine is used to produce hybridization-sensitive fluorescent DNA probes with self-avoidance ability. |
| Ribitol
Ribitol (Adonitol) is a crystalline pentose alcohol formed by the reduction of ribose. |
| Glycodeoxycholic Acid
Glycodeoxycholic Acid is an endogenous metabolite. Glycodeoxycholic Acid induces hepatocyte necrosis and autophagy in patients with obstructive cholestasis. |
| Glycolithocholic acid
Glycolithocholic acid, an endogenous metabolite, is a glycine conjugated secondary bile acid. Glycolithocholic acid can be used to study ulcerative colitis (UC), non-alcoholic steatohepatitis (NASH0) and primary sclerosing cholangitis (PSC). |
| 4-Aminohippuric Acid
4-Aminohippuric acid is a typical substrate of organic anion transport systems. 4-Aminohippuric acid is a diagnostic agent, useful in medical tests involving the kidney, used in the measurement of renal plasma flow. |
All AbMole products are for research use only, cannot be used for human consumption or veterinary use. We do not provide products or services to individuals. Please comply with the intended use and do not use AbMole products for any other purpose.


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2026 AbMole BioScience. All Rights Reserved.
